Welcome to Lakeview TMS Center!
Providing Safe and Effective NeuroStar Advanced TMS Therapy
At Lakeview TMS Center, we offer NeuroStar® Advanced TMS Therapy, an FDA-cleared, non-invasive treatment for:
- Depression
- Anxious depression
- Obsessive-compulsive disorder (OCD)
TMS uses targeted magnetic pulses to stimulate brain networks involved in mood regulation and is clinically proven to improve symptoms in patients who have not responded to antidepressant medications—without the common side effects associated with medication.
NeuroStar TMS Therapy is:
- FDA-cleared and clinically proven
- Non-invasive and drug-free
- Safe and well-tolerated
- Free of long-term systemic side effects
- For patients who prefer or need a drug-free treatment.
Schedule a Complimentary Consultation

Healing isn’t linear, and progress doesn’t always look the same for everyone. Small steps, consistency, and support all matter.
Our office will be closed on:
Memorial Day (May 25th, 2026)
Fourth Of July (July 4th, 2026)
Due to inclement weather our office will be closed Thursday 1/29/26 for all in person appointments/ meetings.
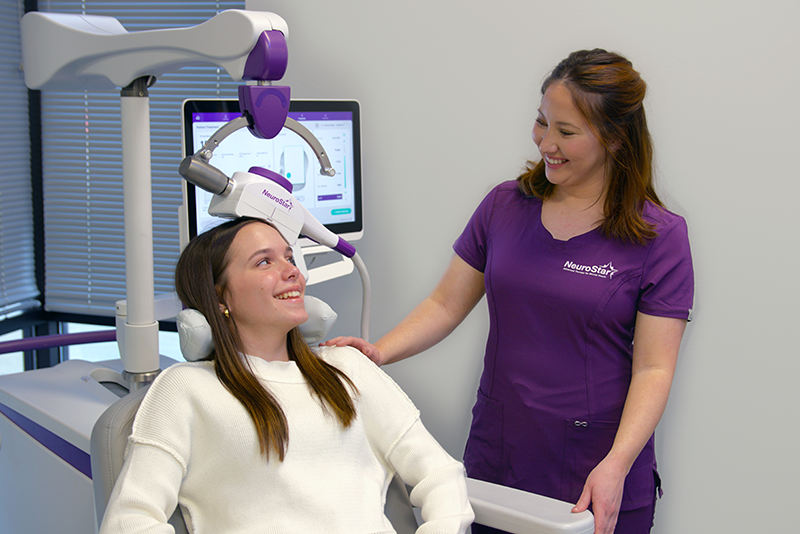
NeuroStar is now FDA-cleared for the treatment of depression in adolescents age 15 and above!
- TMS is also FDA-cleared for the Treatment of OCD and Anxious Depression

NeuroStar Advanced TMS Therapy is The Right Choice
Transcranial magnetic stimulation (TMS) uses a targeted pulsed magnetic field, similar to what is used in an MRI (magnetic resonance imaging) machine. While the patient is awake and alert, NeuroStar Advanced TMS Therapy stimulates areas of the brain that are underactive in depression.
About TMS Therapy
Drug free and noninvasive
Typical treatment consists of 5 sessions per week over 7 weeks
Each treatment typically lasts approximately 20 minutes
Patients can read or watch TV and return to work after

TMS Therapy Media Coverage
- 04/21/2026 Optum Expands TMS Policy to Include Nurse Practitioners
- 01/27/2026 Magnetic pulses to the brain emerge as low-cost lifeline for depression
- 11/20/2025 TRICARE Expands TMS Coverage for Teens as Neuronetics Gains Major Policy Win
- 09/17/2025 New York State Medicaid Expands Coverage for TMS Therapy, Including NeuroStar® Advanced Therapy, to Treat Major Depressive Disorder (MDD)
- 08/22/2025 Neuronetics Announces Expanded Coverage for NeuroStar TMS to Treat Adolescent Depression
What Patients are Saying
“Such a blessing for me and everyone who loves me to see improvements start here, and I am still getting better! For me, it has been an answer to prayers!”
HG
“By the end of the TMS treatment, things were better than I could have imagined. After that sixth week, I was so excited that my life was starting to resume.”


